Understanding high and low factor activity levels, peaks and troughs, and half-life
Factor VIII is a vital blood-clotting protein that is deficient or defective in individuals with hemophilia A. Its primary role is to work with other clotting factors to form a blood clot. Understanding the ins and outs of Factor VIII activity levels can help patients better communicate with their healthcare providers and effectively manage their hemophilia.
Understanding elevated, normal, and decreased Factor VIII activity levels
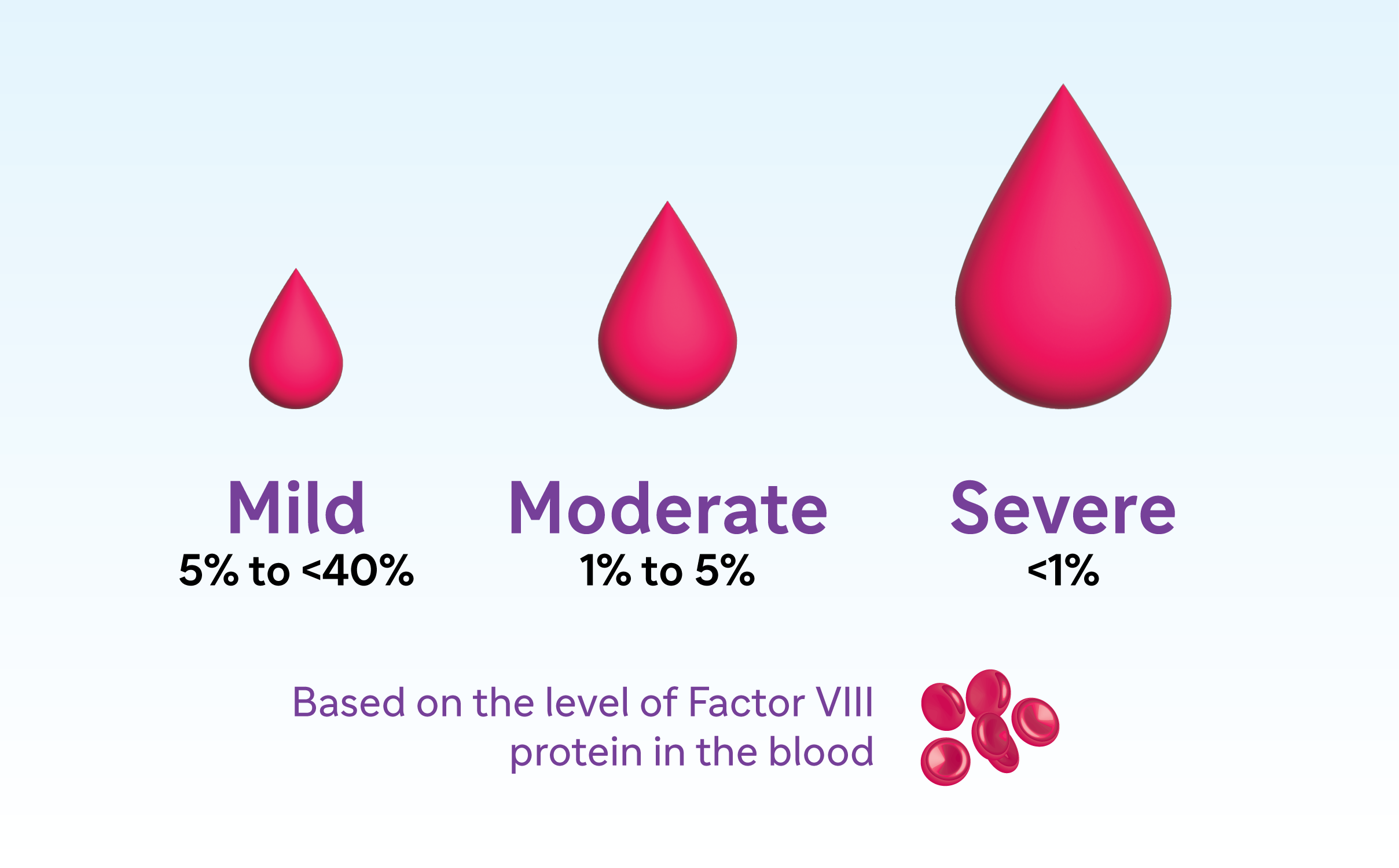
Patients with hemophilia may have Factor VIII activity anywhere between 1%-<40% and are accordingly divided into 3 disease categories – mild (5%-<40%), moderate (1%-5%), or severe (<1%). Maintenance of Factor VIII activity at an optimal level can help patients manage their hemophilia and control bleeding.
Impact of low or decreased Factor VIII activity levels in hemophilia
Low or decreased Factor VIII activity levels can lead to an increased risk of bleeding, particularly in joints and muscles. Managing Factor VIII levels is essential to reduce the risk of bleeding and maintain joint health in patients with hemophilia.
Changes in Factor VIII levels during pregnancy
In women who are hemophilia carriers, Factor VIII levels may increase during pregnancy. However, the Factor VIII level may again become low after delivery, posing a risk for severe bleeding known as postpartum hemorrhage, requiring treatment to stop bleeding.

Importance of monitoring Factor VIII
activity levels
Patients should work with their doctor to measure their Factor VIII levels to understand changes in their levels. It is particularly important for patients on prophylactic treatment to ensure that their treatment is effective and appropriately adjusted. During prophylactic treatment with factor replacement therapies, clotting factors are regularly infused to prevent bleeding episodes from occurring. People with hemophilia need to work with their healthcare providers for treatment plans, both short- and long-term.
Understanding peak and trough levels
When people with hemophilia receive a Factor VIII infusion, the amount of the Factor VIII protein in their blood initially increases, followed by a steady decrease in levels, also referred to as peaks (highs) and troughs (lows).
Half-life of Factor VIII and its impact on hemophilia treatment
Half-life is the length of time that it takes for only half of a drug to remain active in a patient’s body. The half-life of Factor VIII varies among patients and can be influenced by age, presence of inhibitors, and the specific Factor VIII products that are used in the treatment. Understanding a drug’s half-life plays a key role in determining the appropriate dosing regimen for a treatment.
Impact on Factor VIII infusion
The half-life of Factor VIII affects the timing and dosage of replacement therapy. Factor replacement therapy is a treatment for hemophilia A where patients receive infusions of clotting Factor VIII to help their blood clot properly and prevent bleeding. A personalized understanding of half-life can lead to a more effective and less burdensome treatment plan for patients.
Frequently asked questions on Factor VIII activity levels
Find your CoRe Manager and connect today!
Sanofi Hemophilia Community Relations and Education (CoRe) Managers offer education to
people living with hemophilia and their families. CoRe Managers provide information about
living with hemophilia and treatment options. Use our handy CoRe Locator to find the CoRe
team member nearest you.
ALTUVIIIO® [antihemophilic factor (recombinant), Fc-VWF-XTEN fusion protein-ehtl] is an injectable medicine that is used to control and reduce the number of bleeding episodes in people with hemophilia A (congenital Factor VIII deficiency).
Your healthcare provider may give you ALTUVIIIO when you have surgery.
What is the most important information I need to know about ALTUVIIIO?
Do not attempt to give yourself an injection unless you have been taught how by your healthcare provider or hemophilia center. You must carefully follow your healthcare provider’s instructions regarding the dose and schedule for injecting ALTUVIIIO so that your treatment will work best for you.
Who should not use ALTUVIIIO?
You should not use ALTUVIIIO if you have had an allergic reaction to it in the past.
What should I tell my healthcare provider before using ALTUVIIIO?
Tell your healthcare provider if you have had any medical problems, take any medications, including prescription and non-prescription medicines, supplements, or herbal medicines, are breastfeeding, or are pregnant or planning to become pregnant.
What are the possible side effects of ALTUVIIIO?
You can have an allergic reaction to ALTUVIIIO. Call your healthcare provider or emergency department right away if you have any of the following symptoms: difficulty breathing, chest tightness, swelling of the face, rash, or hives.
Your body can also make antibodies called “inhibitors” against ALTUVIIIO. This can stop ALTUVIIIO from working properly. Your healthcare provider may give you blood tests to check for inhibitors.
The common side effects of ALTUVIIIO are headache and joint pain.
These are not the only possible side effects of ALTUVIIIO. Tell your healthcare provider about any side effect that bothers you or does not go away.
Please see full Prescribing Information.
Learn more about Sanofi’s commitment to fighting counterfeit drugs.
![ALTUVIIIO® [Antihemophilic Factor (Recombinant), Fc-VWF-XTEN Fusion Protein-ehtl] logo](/.imaging/default/dam/Marketing/AltuviiioUS/altuviiio.webp/jcr:content.webp)