

Watch how ALTUVIIIO® [antihemophilic factor (recombinant), Fc-VWF-XTEN fusion protein-ehtl] works
Hemophilia A is a rare, inherited blood disorder caused by an imbalance in hemostasis, a tightly controlled process that forms a clot to stop a bleed, due to missing or deficient Factor VIII.
In healthy people, platelets are first recruited to a site of injury and, with the help of von Willebrand factor, a “platelet plug” forms to reduce blood loss.
A multistep process, known as the clotting cascade, is then set into motion. A series of clotting factors, such as Factor VIII, are activated, the protein thrombin is produced and turns into fibrin to help create a mesh around the platelet plug to stabilize the clot.
In hemophilia A, clotting Factor VIII is missing or not working, which prevents clot formation and results in excessive bleeding.
Current treatments for hemophilia A include infusions of replacement Factor VIII to prevent bleeds or treat them when they occur.
Once in the bloodstream, Factor VIII binds to von Willebrand factor. And when von Willebrand factor is cleared from the blood, Factor VIII goes along with it.
This has limited the amount of time Factor VIII replacement therapies were able to stay in the blood, usually requiring multiple infusions a week.
ALTUVIIIO® is the first and only Factor VIII replacement therapy of its kind designed to help Factor VIII stay in the body for longer.
In a Phase 3 study, ALTUVIIIO offered the longest half-life of any Factor VIII replacement therapy: a 48-hour half-life in adults. The half-life was 44.6 hours for adolescents aged 12 years to under 18 years, 42.4 hours for children aged 6 years to under 12 years, and 38 hours for children aged 1 year to under 6 years after one infusion.
ALTUVIIIO uniquely combines 3 components to help it stay in your body longer.
One. The Factor VIII in ALTUVIIIO is already bound to fragments of von Willebrand factor, keeping it from binding to natural von Willebrand factor in the bloodstream. This helps to sustain ALTUVIIIO in the body for longer.
Two. XTEN proteins also protect ALTUVIIIO from breaking down too early.
Three. Fc fusion helps to redirect ALTUVIIIO back into the bloodstream, preventing it from being broken down inside the cell and allowing it to circulate for longer.
All 3 components work together to keep ALTUVIIIO Factor VIII levels higher for longer. In a Phase 1 study, ALTUVIIIO had a 3- to 4-times longer half-life than extended half-life and standard half-life therapies.
ALTUVIIIO also stayed above 40% in the normal to near-normal range for most of the week with one infusion in adults.
When a bleed occurs, the ALTUVIIIO molecule releases the XTEN chains and the von Willebrand factor fragments, releasing ALTUVIIIO to work in the clotting cascade.
The protein thrombin is produced and turns into fibrin to promote clotting and stop the bleed.
Talk to your doctor about making the switch to Factor Up with ALTUVIIIO.
INDICATION
ALTUVIIIO® [antihemophilic factor (recombinant), Fc-VWF-XTEN fusion protein-ehtl] is an injectable medicine that is used to control and reduce the number of bleeding episodes in people with hemophilia A (congenital Factor VIII deficiency).
Your healthcare provider may give you ALTUVIIIO when you have surgery.
IMPORTANT SAFETY INFORMATION
What is the most important information I need to know about ALTUVIIIO?
Do not attempt to give yourself an injection unless you have been taught how by your healthcare provider or hemophilia center. You must carefully follow your healthcare provider's instructions regarding the dose and schedule for injecting ALTUVIIIO so that your treatment will work best for you.
Who should not use ALTUVIIIO?
You should not use ALTUVIIIO if you have had an allergic reaction to it in the past.
What should I tell my healthcare provider before using ALTUVIIIO?
Tell your healthcare provider if you have had any medical problems, take any medications, including prescription and non-prescription medicines, supplements, or herbal medicines, are breastfeeding, or are pregnant or planning to become pregnant.
What are the possible side effects of ALTUVIIIO?
You can have an allergic reaction to ALTUVIIIO. Call your healthcare provider or emergency department right away if you have any of the following symptoms: difficulty breathing, chest tightness, swelling of the face, rash, or hives.
Your body can also make antibodies called “inhibitors” against ALTUVIIIO. This can stop ALTUVIIIO from working properly. Your healthcare provider may give you blood tests to check for inhibitors.
The common side effects of ALTUVIIIO are headache and joint pain.
These are not the only possible side effects of ALTUVIIIO. Tell your healthcare provider about any side effect that bothers you or does not go away.
Please see full Prescribing Information.
The unique design of ALTUVIIIO keeps factor levels higher for longer
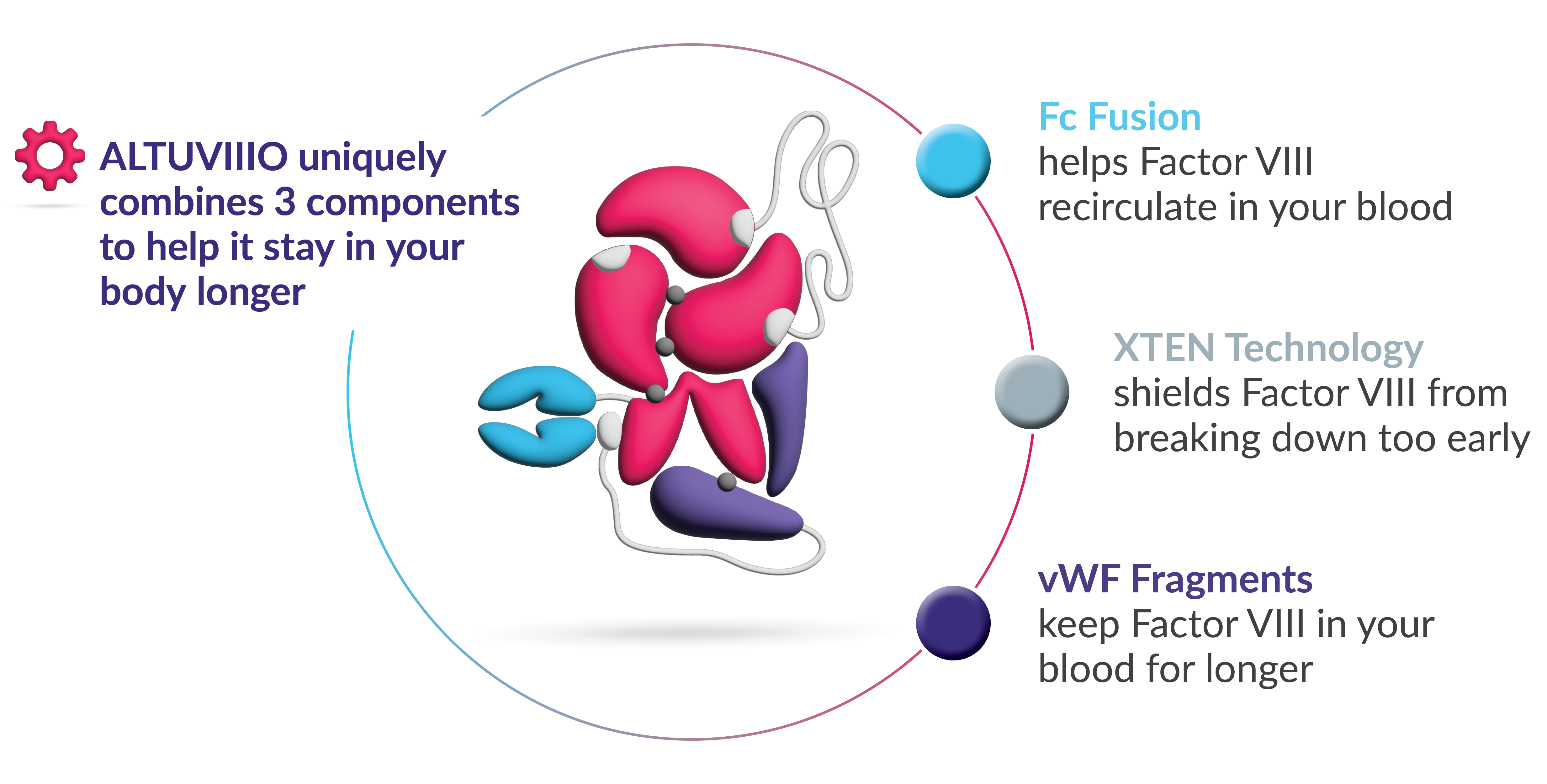
How exactly does ALTUVIIIO work in the body?
ALTUVIIIO uniquely combines 3 components to help it stay in your body longer: Fc region of IgG, XTEN technology, and vWF fragments. All 3 components work together to release rFVIII so it can stop the bleed.
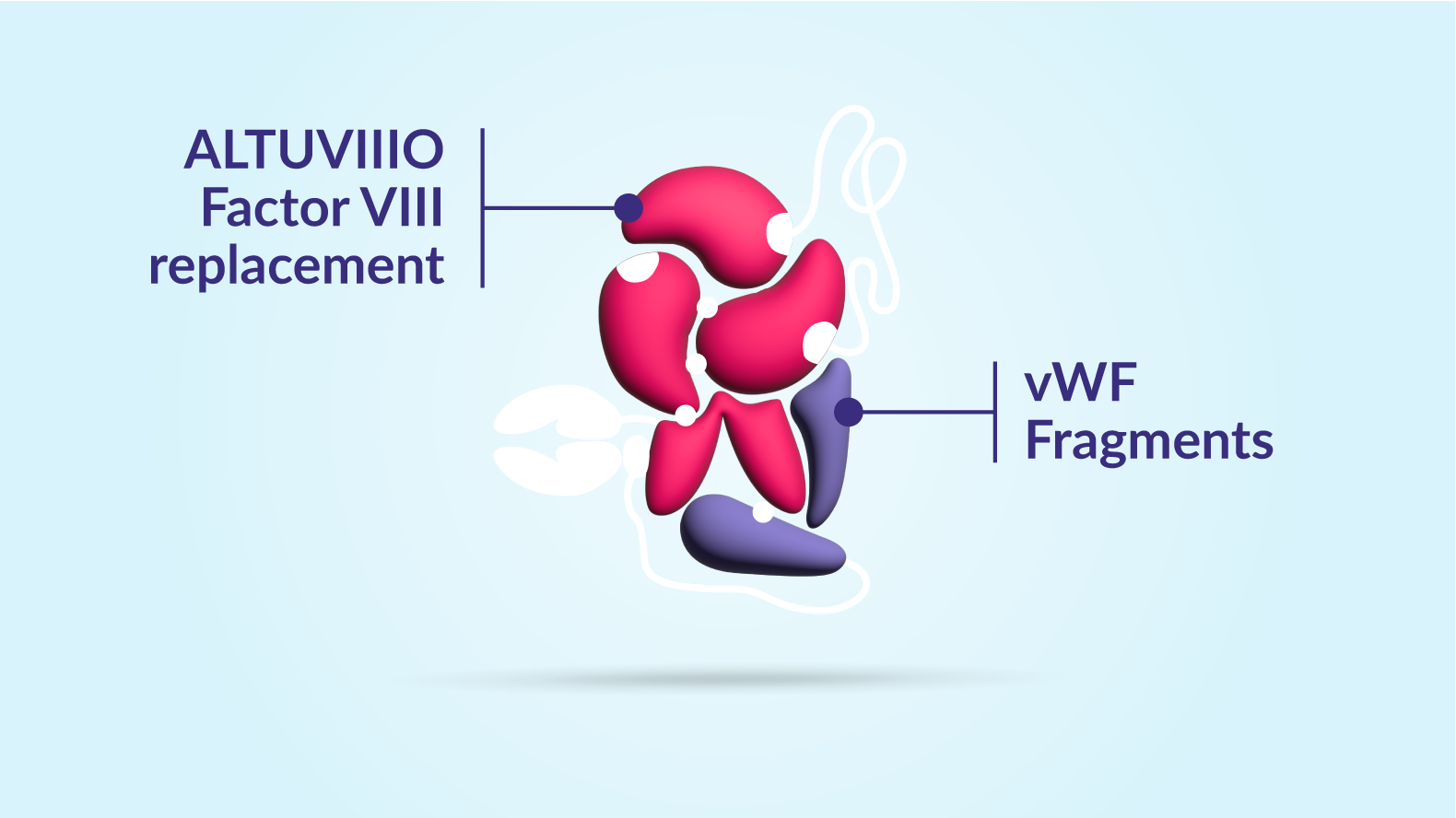
Sustain
The Factor VIII in ALTUVIIIO is already bound to fragments of von Willebrand factor, keeping it from binding to natural von Willebrand factor in the bloodstream. This helps to sustain ALTUVIIIO in the body for longer.
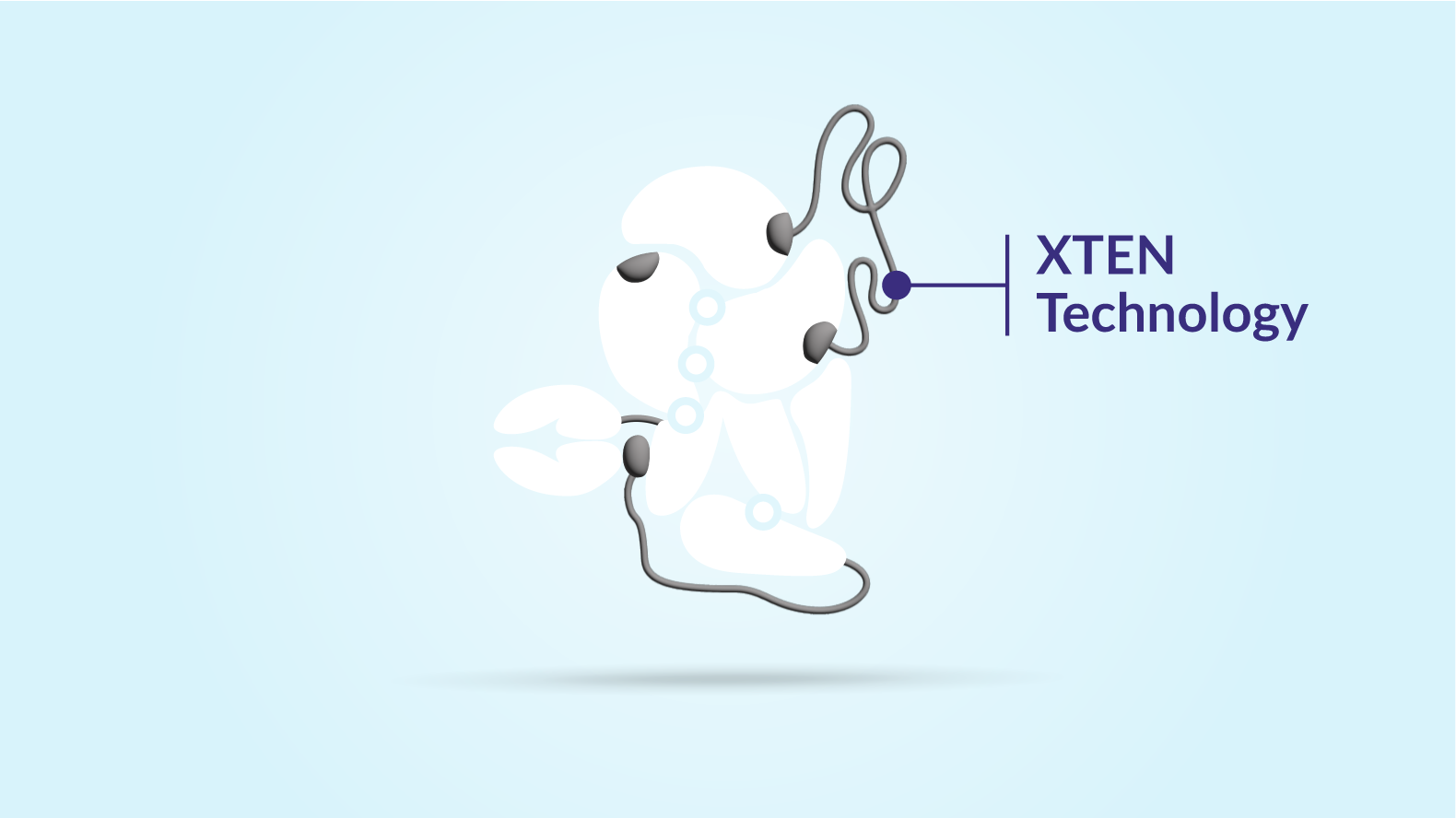
Protect
XTEN technology also protects ALTUIIIO from being broken down. When a bleed occurs, the XTEN chains release the von Willebrand factor fragments, activating ALTUVIIIO to promote clotting.
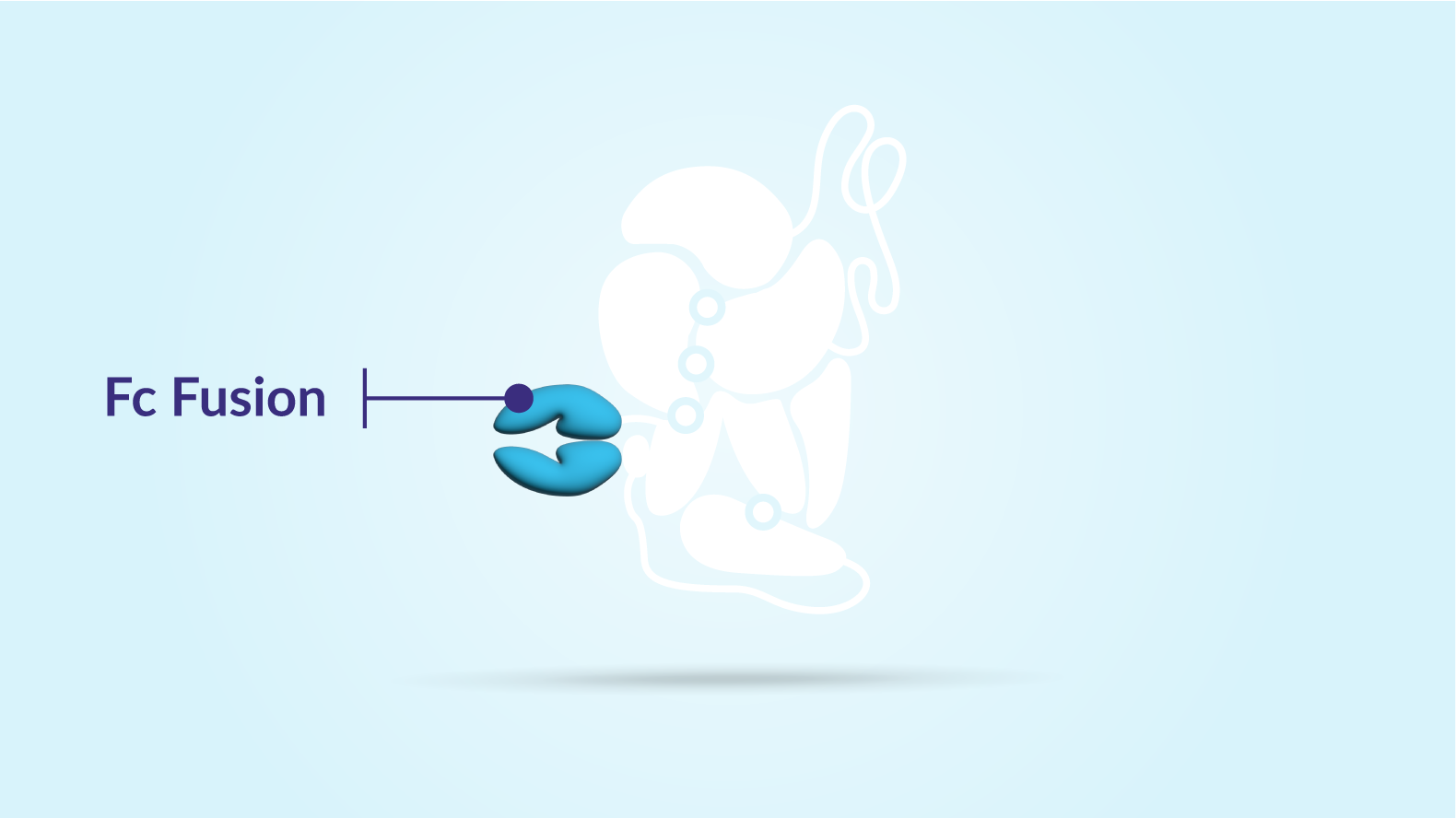
Redirect
The Fc protein helps to redirect ALTUVIIIO back into the bloodstream, preventing it from being broken down inside the cell and allowing it to circulate for longer.
IgG=immunoglobulin G; rFVIII=recombinant Factor VIII.
Find your CoRe Manager and connect today!
Sanofi Hemophilia Community Relations and Education (CoRe) Managers offer education to
people living with hemophilia and their families. CoRe Managers provide information about
living with hemophilia and treatment options. Use our handy CoRe Locator to find the CoRe
team member nearest you.
ALTUVIIIO® [antihemophilic factor (recombinant), Fc-VWF-XTEN fusion protein-ehtl] is an injectable medicine that is used to control and reduce the number of bleeding episodes in people with hemophilia A (congenital Factor VIII deficiency).
Your healthcare provider may give you ALTUVIIIO when you have surgery.
What is the most important information I need to know about ALTUVIIIO?
Do not attempt to give yourself an injection unless you have been taught how by your healthcare provider or hemophilia center. You must carefully follow your healthcare provider’s instructions regarding the dose and schedule for injecting ALTUVIIIO so that your treatment will work best for you.
Who should not use ALTUVIIIO?
You should not use ALTUVIIIO if you have had an allergic reaction to it in the past.
What should I tell my healthcare provider before using ALTUVIIIO?
Tell your healthcare provider if you have had any medical problems, take any medications, including prescription and non-prescription medicines, supplements, or herbal medicines, are breastfeeding, or are pregnant or planning to become pregnant.
What are the possible side effects of ALTUVIIIO?
You can have an allergic reaction to ALTUVIIIO. Call your healthcare provider or emergency department right away if you have any of the following symptoms: difficulty breathing, chest tightness, swelling of the face, rash, or hives.
Your body can also make antibodies called “inhibitors” against ALTUVIIIO. This can stop ALTUVIIIO from working properly. Your healthcare provider may give you blood tests to check for inhibitors.
The common side effects of ALTUVIIIO are headache and joint pain.
These are not the only possible side effects of ALTUVIIIO. Tell your healthcare provider about any side effect that bothers you or does not go away.
Please see full Prescribing Information.
Learn more about Sanofi’s commitment to fighting counterfeit drugs.
![ALTUVIIIO® [Antihemophilic Factor (Recombinant), Fc-VWF-XTEN Fusion Protein-ehtl] logo](/.imaging/default/dam/Marketing/AltuviiioUS/altuviiio.webp/jcr:content.webp)