Clinical data in kids 1 year to <12 years
After the first dose of ALTUVIIIO® [antihemophilic factor (recombinant), Fc-VWF-XTEN fusion protein-ehtl], factor activity levels were measured in a group of 18 children aged 6 to <12 years in the XTEND-Kids study
Factor activity levels for children aged 6 to <12 years
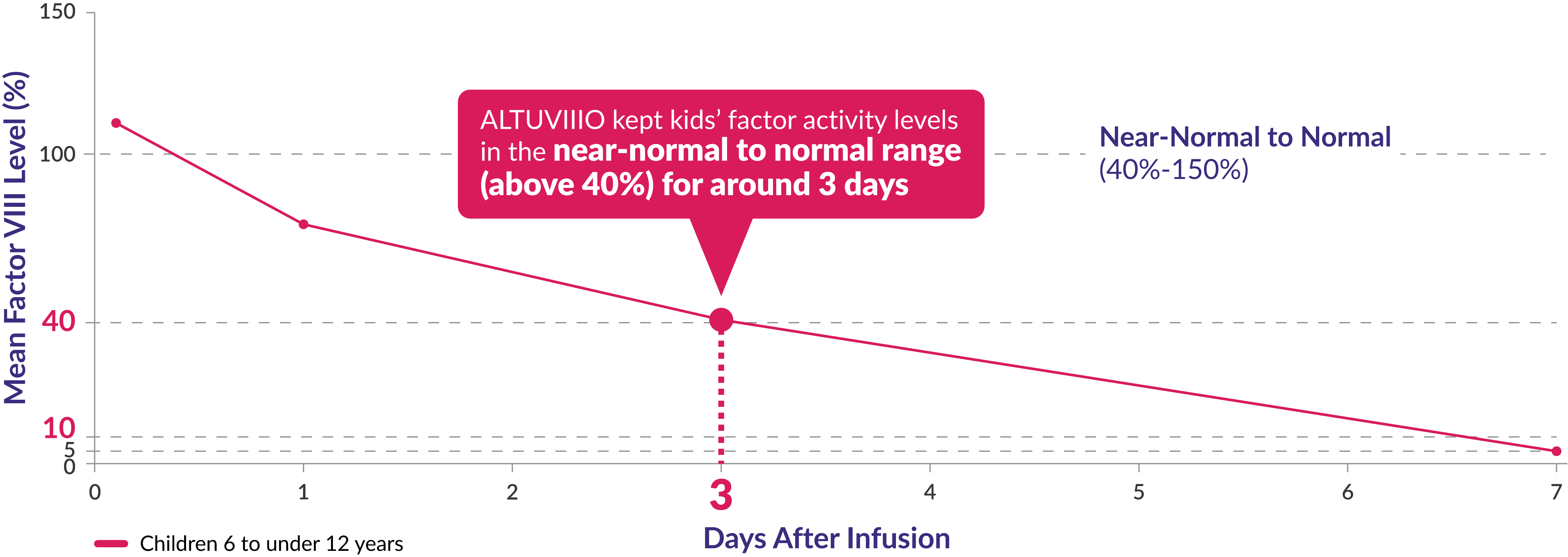
After one year on ALTUVIIIO prophylaxis, average Factor VIII trough levels were 17% in children 6 to ‹12 years*
*Evaluated in 36 children aged 6 years to <12 years in the XTEND-Kids study.
After the first dose of ALTUVIIIO, factor activity levels were measured in a group of 19 children aged <6 years in the XTEND-Kids study
Factor activity levels for children aged <6 years
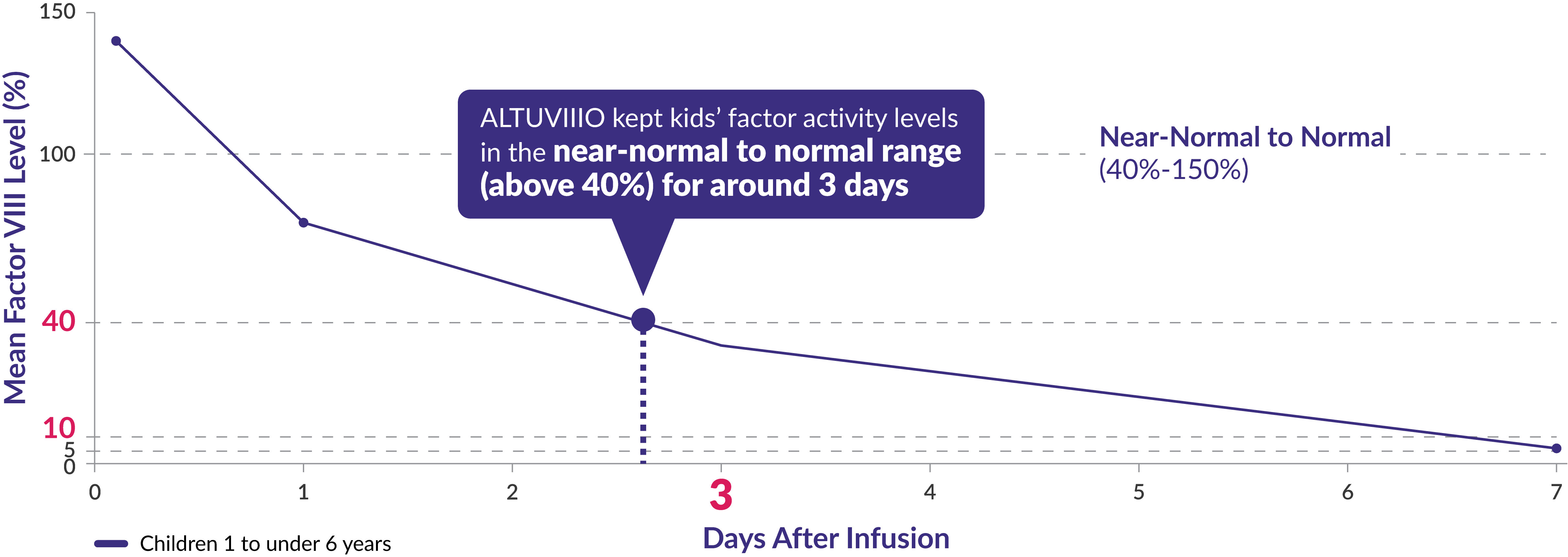
After one year on ALTUVIIIO prophylaxis, average Factor VIII trough levels were 11% in children 1 to ‹6 years†
†Evaluated in 38 children aged 1 year to <6 years in the XTEND-Kids study.
What do the results mean?
- Peaks refer to the highest amount of factor in your child’s body after infusion
- Troughs are the lowest levels of factor before the next infusion
- Half-life is the length of time that it takes for only half the drug to remain active in your body. A drug's half-life plays a key role in dosing
ALTUVIIIO has the longest half-life of any Factor VIII replacement therapy for children with hemophilia A.‡

ALTUVIIIO was studied over 1 year in the XTEND-Kids study
74 previously treated male children under 12 years of age who switched to ALTUVIIIO:
- 36 children aged 6 to under 12 years
- 38 children under the age of 6
- Efficacy was evaluated in 72 of these children
The primary goal of the XTEND-Kids study was to determine whether or not children under 12 years of age developed inhibitors to ALTUVIIIO.
Although no inhibitors were found in clinical studies, inhibitors have occurred with ALTUVIIIO in the postmarketing setting.

Kids can experience proven bleed protection on ALTUVIIIO prophylaxis§

Median bleeds per year||
(median annualized bleed rate)

Mean bleeds per year||
(mean annualized bleed rate)

Median joint bleeds per year||
(median annualized joint bleed rate)
Over one year on ALTUVIIIO prophylaxis
of children had ZERO bleeding episodes
of children had ZERO joint bleeds
of children had ZERO spontaneous bleeds
‡The half-life was 42.4 hours for children aged 6 to under 12 years and 38 hours for children aged 1 to under 6 years.
§Efficacy was evaluated in 72 children in the XTEND-Kids study.
||Data based on treated bleeds.
Find your CoRe Manager and connect today!
Sanofi Hemophilia Community Relations and Education (CoRe) Managers offer education to
people living with hemophilia and their families. CoRe Managers provide information about
living with hemophilia and treatment options. Use our handy CoRe Locator to find the CoRe
team member nearest you.
ALTUVIIIO® [antihemophilic factor (recombinant), Fc-VWF-XTEN fusion protein-ehtl] is an injectable medicine that is used to control and reduce the number of bleeding episodes in people with hemophilia A (congenital Factor VIII deficiency).
Your healthcare provider may give you ALTUVIIIO when you have surgery.
What is the most important information I need to know about ALTUVIIIO?
Do not attempt to give yourself an injection unless you have been taught how by your healthcare provider or hemophilia center. You must carefully follow your healthcare provider’s instructions regarding the dose and schedule for injecting ALTUVIIIO so that your treatment will work best for you.
Who should not use ALTUVIIIO?
You should not use ALTUVIIIO if you have had an allergic reaction to it in the past.
What should I tell my healthcare provider before using ALTUVIIIO?
Tell your healthcare provider if you have had any medical problems, take any medications, including prescription and non-prescription medicines, supplements, or herbal medicines, are breastfeeding, or are pregnant or planning to become pregnant.
What are the possible side effects of ALTUVIIIO?
You can have an allergic reaction to ALTUVIIIO. Call your healthcare provider or emergency department right away if you have any of the following symptoms: difficulty breathing, chest tightness, swelling of the face, rash, or hives.
Your body can also make antibodies called “inhibitors” against ALTUVIIIO. This can stop ALTUVIIIO from working properly. Your healthcare provider may give you blood tests to check for inhibitors.
The common side effects of ALTUVIIIO are headache and joint pain.
These are not the only possible side effects of ALTUVIIIO. Tell your healthcare provider about any side effect that bothers you or does not go away.
Please see full Prescribing Information.
Learn more about Sanofi’s commitment to fighting counterfeit drugs.
![ALTUVIIIO® [Antihemophilic Factor (Recombinant), Fc-VWF-XTEN Fusion Protein-ehtl] logo](/.imaging/default/dam/Marketing/AltuviiioUS/altuviiio.webp/jcr:content.webp)