

ALTUVIIIO® [antihemophilic factor (recombinant), Fc-VWF-XTEN fusion protein-ehtl] is a first-in-class, Factor VIII replacement therapy that keeps factor activity levels higher for most of the week with one weekly infusion
The only once-weekly prophylaxis factor infusion
3-4x longer half-life, in a separate study with EHL and SHL therapies*
In a Phase 1 study, ALTUVIIIO had a 3-4x longer
half-life than EHL and SHL therapies*
*This is information from a study of 13 previously treated adults with severe hemophilia A, that had the goal of comparing how long ALTUVIIIO, Adynovate® [Antihemophilic Factor (Recombinant), PEGylated], and Advate® [Antihemophilic Factor (Recombinant)] stayed in the body after 1 dose. Half-life was 43 hours for ALTUVIIIO, 15 hours for Adynovate, and 11 hours for Advate. Adynovate and Advate are registered trademarks of Baxalta Incorporated, a Takeda company.
EHL=extended half-life; SHL=standard half-life.
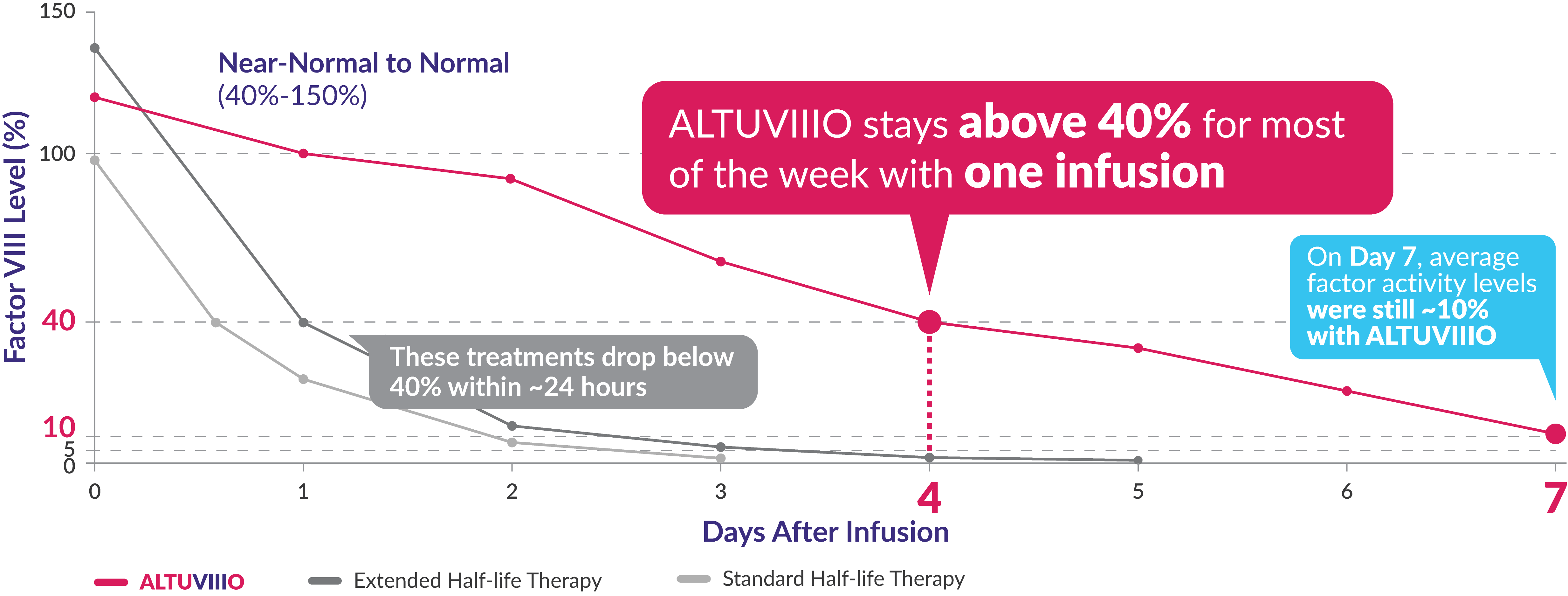
In the XTEND-1 study, average Factor VIII levels stayed above:
For most of the week in adults
( near-normal to normal range )
On average for adults, for the entire week
Average trough levels were 9% for adolescents aged 12 years to under 18 years.
How was ALTUVIIIO studied?
ALTUVIIIO was studied in XTEND-1 for one year

The XTEND-1 study broke patients into 2 different treatment groups: Group 1 (133 people aged 12 years and older) switched from prior prophylaxis therapy to ALTUVIIIO for 52 weeks. Efficacy of prophylaxis was studied in 128 of these patients. Group 2 (26 people aged 12 years and older) switched from prior on-demand therapy to on demand with ALTUVIIIO and later switched to ALTUVIIIO prophylaxis.
Finding people's mean annualized bleed rate was the primary goal of the study.
Proven bleed protection with ALTUVIIIO prophylaxis
Data from the XTEND-1 study

Median bleeds per year*
(median annualized bleed rate)

Mean bleeds per year*
(mean annualized bleed rate)
PRIMARY OUTCOME

Median joint bleeds per year*
(median annualized joint bleed rate)
How were bleeds and joint bleeds measured in the trials?
- Median ABR was the middle number of all ABRs, when everyone’s ABR was ordered from least to greatest
- Mean ABR was the average number based on everyone’s ABR
Significant improvement in bleed protection with ALTUVIIIO prophylaxis
In Group 1:
133 people aged 12 years and older had prior prophylaxis therapy and switched to ALTUVIIIO.
78 of those people participated in a separate study to measure their ABRs on their prior
prophylaxis. Comparing the results of the 78 people who participated in both studies showed:
On average, people went from 3 bleeds to less than 1 bleed a year (mean ABR 0.7). That’s a 77% reduction in yearly bleeds (mean).*
And, on average, over 52 weeks on prophylaxis, 64% of people had 0 bleeds* vs 42% on prior prophylaxis.
*Data based on treated bleeds.
ABR=annualized bleed rate.
Know your joints are protected

Joint health score
- A mean change of -1.5 (-2.7, -0.3) from starting point was observed in Hemophilia Joint Health Score (HJHS) total score
- HJHS is a validated tool used to measure joint health function. The study was designed so the investigators knew the patients were taking ALTUVIIIO, which may have impacted their assessment of the patients’ HJHS score

100% Target joint resolution
- Target joints are 3 or more spontaneous bleeds in a major joint in a consecutive 6-month period
- Target joints were considered resolved if 2 or fewer bleeds occurred in a target joint in 12 months
*Data based on treated bleeds.
Demand fewer yearly bleeds
Switch it up from on demand to prophylaxis with ALTUVIIIO.
In Group 2:
26 people aged 12 years and older were treated on demand with ALTUVIIIO, then switched to ALTUVIIIO prophylaxis for 26 weeks.
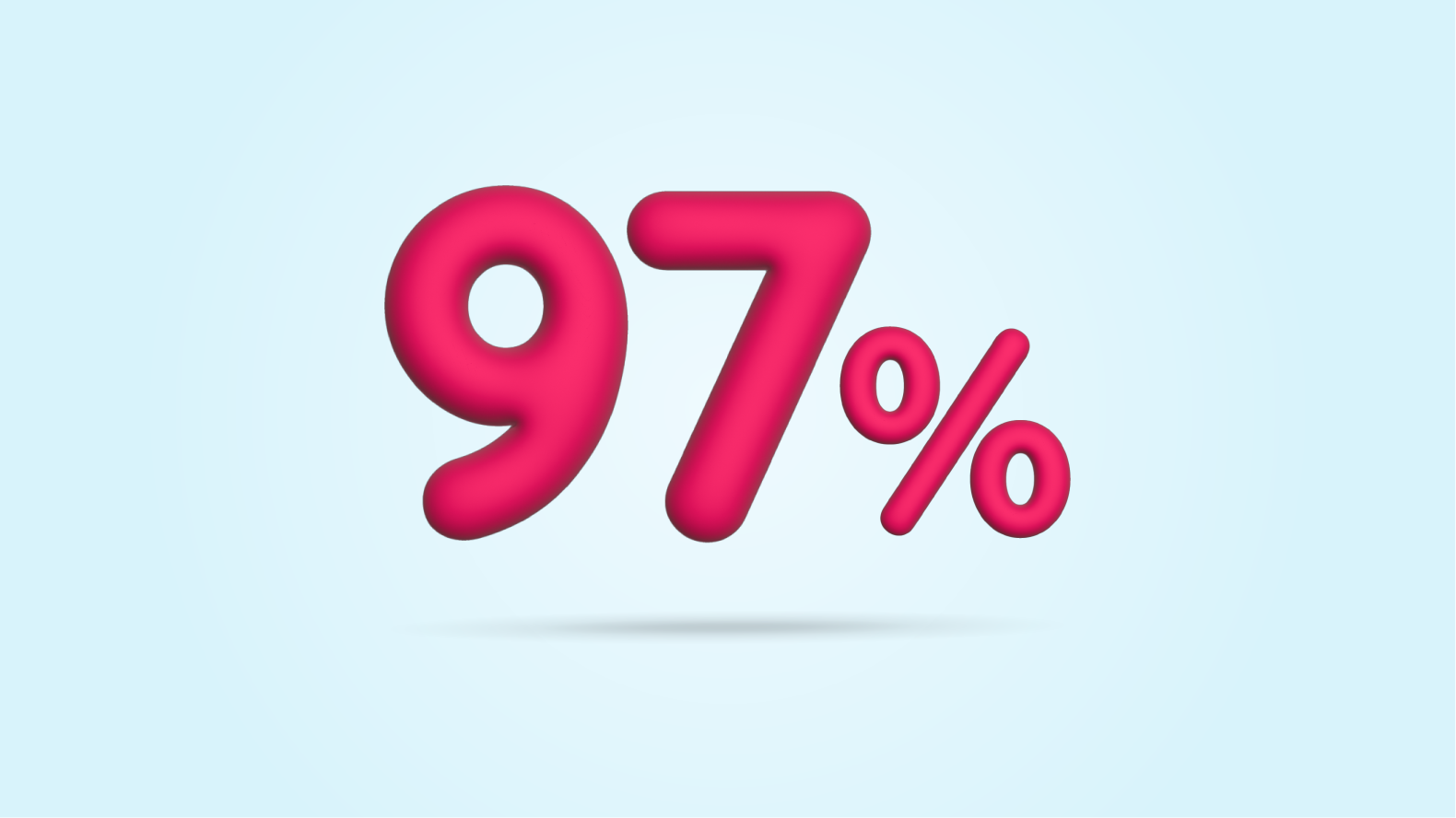
On average, people who switched from ALTUVIIIO on demand to ALTUVIIIO prophylaxis went from 21 bleeds to less than 1 bleed a year (mean ABR 0.7). That’s a 97% reduction in yearly bleeds (mean).*
And, over 26 weeks on prophylaxis, 77% of people had 0 bleeds* and 81% of people had 0 joint bleeds.*
*Data based on treated bleeds.
ABR=annualized bleed rate.
Pain and physical health from patient-reported outcomes
In people on once-weekly ALTUVIIIO who switched from prior prophylaxis in XTEND-1
Physical health
Change observed in Haem-A-QoL Physical Health Score from 37.0 at baseline to 29.7 at Week 52.*
Pain intensity
Reduction observed in PROMIS Pain Intensity 3a Score from 2.5 at
baseline to 2.2 at Week 52.*
A lower score represents an overall improvement in these measures. The XTEND-1 study was
designed so patients and their doctors knew they were taking ALTUVIIIO, which may have impacted these findings. The study was a single-arm study so all participants in the trial were treated with ALTUVIIIO and there was no other treatment to compare, which may impact the ability to assess the effect of ALTUVIIIO on these patient-reported outcomes. The PROMIS tool was not specifically developed for use in hemophilia patients.
*Patient-reported outcomes of pain intensity and physical-health scores were evaluated in patients receiving ALTUVIIIO prophylaxis in Group 1. The PROMIS Pain intensity 3a instrument was used to assess pain, specifically the first question that rates the worst pain experienced during the last 7 days. Physical-health scores were assessed in patients 17 years and older using the Physical Health Score of Haem-A-QoL, which evaluated factors such as painful swellings, presence of joint pain, pain with movement, difficulty walking far, and needing more time to get ready.
Haem-A-QoL=Haemophilia Quality of Life Questionnaire for Adults; PROMIS=Patient-Reported Outcome Measurement Information System.
One infusion, a powerful* response
Bleed control with ALTUVIIIO
In the clinical trial of 362 bleeds that occurred, 97% were treated with one
infusion.†
*Based on the number of infusions needed to treat an active bleed.
†In the XTEND-1 study, 350 out of 362 bleeds that occurred were resolved with only one infusion.
Find your CoRe Manager and connect today!
Sanofi Hemophilia Community Relations and Education (CoRe) Managers offer education to
people living with hemophilia and their families. CoRe Managers provide information about
living with hemophilia and treatment options. Use our handy CoRe Locator to find the CoRe
team member nearest you.
ALTUVIIIO® [antihemophilic factor (recombinant), Fc-VWF-XTEN fusion protein-ehtl] is an injectable medicine that is used to control and reduce the number of bleeding episodes in people with hemophilia A (congenital Factor VIII deficiency).
Your healthcare provider may give you ALTUVIIIO when you have surgery.
What is the most important information I need to know about ALTUVIIIO?
Do not attempt to give yourself an injection unless you have been taught how by your healthcare provider or hemophilia center. You must carefully follow your healthcare provider’s instructions regarding the dose and schedule for injecting ALTUVIIIO so that your treatment will work best for you.
Who should not use ALTUVIIIO?
You should not use ALTUVIIIO if you have had an allergic reaction to it in the past.
What should I tell my healthcare provider before using ALTUVIIIO?
Tell your healthcare provider if you have had any medical problems, take any medications, including prescription and non-prescription medicines, supplements, or herbal medicines, are breastfeeding, or are pregnant or planning to become pregnant.
What are the possible side effects of ALTUVIIIO?
You can have an allergic reaction to ALTUVIIIO. Call your healthcare provider or emergency department right away if you have any of the following symptoms: difficulty breathing, chest tightness, swelling of the face, rash, or hives.
Your body can also make antibodies called “inhibitors” against ALTUVIIIO. This can stop ALTUVIIIO from working properly. Your healthcare provider may give you blood tests to check for inhibitors.
The common side effects of ALTUVIIIO are headache and joint pain.
These are not the only possible side effects of ALTUVIIIO. Tell your healthcare provider about any side effect that bothers you or does not go away.
Please see full Prescribing Information.
Learn more about Sanofi’s commitment to fighting counterfeit drugs.
![ALTUVIIIO® [Antihemophilic Factor (Recombinant), Fc-VWF-XTEN Fusion Protein-ehtl] logo](/.imaging/default/dam/Marketing/AltuviiioUS/altuviiio.webp/jcr:content.webp)